***Sponsored by LFG Equities Corp.

Kadimastem Shareholders Approved the Merger with NLS Pharmaceutics
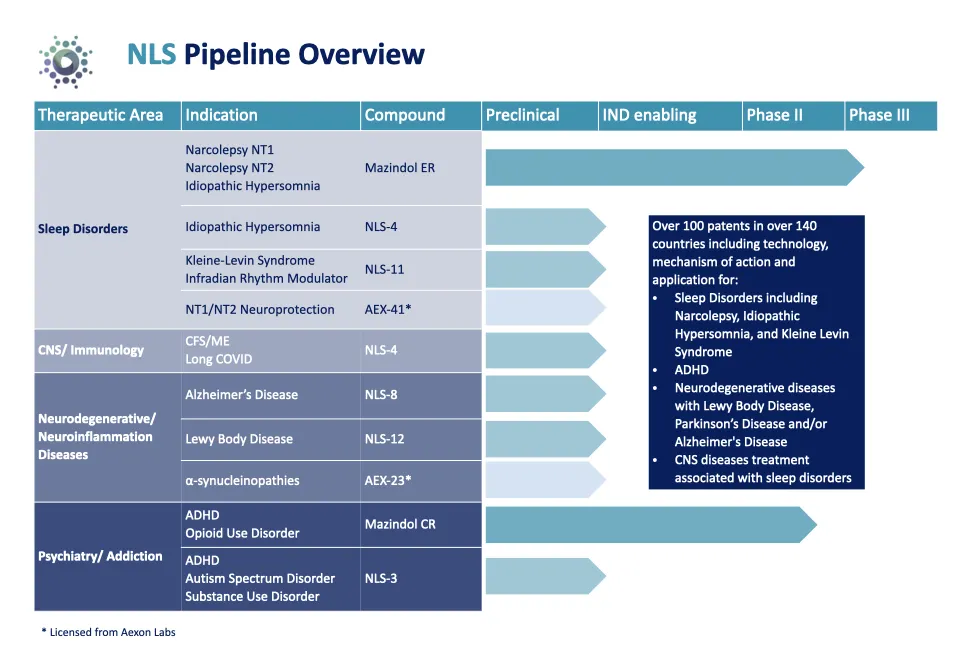
Pipeline strength: Over 100 patents in 140+ countries covering CNS, ADHD, cancer fatigue, Parkinson’s, and more
READ THE INVESTOR PRESENTATION HERE
Exciting news! 🎉 The @SECGov has declared effective our Form F-4 for the planned merger with @Kadimastem.
— NLS Pharmaceutics (@NLSPharma) September 10, 2025
The combined company, NewCelX, will unite cell therapy & small-molecule innovation to tackle #ALS, #Diabetes, & #CNS disorders.https://t.co/KOp6YWfR34 pic.twitter.com/0bBmZfd4fz
_________________
Hello Everyone,
We have a past winner back in the cross hairs ahead of Tuesday’s session.
We have been covering this company for over two years now and have put it in front of you on 3-4 occasions, some with tremendous success.
This one has a history of moving and there have been quite a few developments since we last tasked you with researching NLSP.
The story of NLS is rapidly becoming the story of NewcelX: a debt-free, Nasdaq-listed biotech advancing treatments for ALS, diabetes, addiction, and CNS disorders. With strong financing, validated science, and a merger that multiplies potential, the company is positioned at a powerful inflection point.
This isn’t just a cosmetic change—it’s the signal of a new strategy, new growth opportunities, and a sharper focus on the future.
NLSP is a clinical-stage pharmaceutical company developing innovative therapies for patients with rare and complex central nervous system (CNS) disorders — particularly in areas where existing treatments are insufficient or non-existent. Earlier this year they announced a definitive merger agreement with Kadimastem Ltd. (TASE: KDST) (“Kadimastem”), a clinical-stage cell therapy company specializing in “off-the-shelf” allogeneic cell products for neurodegenerative diseases and diabetes.
The merger will create a company with complementary approaches in cell therapy and small molecules, broadening the therapeutic scope in CNS disorders and metabolic diseases.
The merger is significant because it offers a pathway to greater scale, diversification, capital-market access, and potentially higher valuation.
Uniting NLS’s small molecule / CNS expertise with Kadimastem’s cell therapy pipeline creates a massive vision for their pipeline and current operations.
One of the benefits of the merger is that the new company with have a more diversified and more compelling pipeline. The merger combines cell therapy (Kadimastem) and small molecule / CNS modalities (NLS). This diversification may reduce dependency on any single program. f one program underperforms, the other’s success may balance downside risk. A richer, stronger pipeline makes the company more attractive to investors, partners and potential acquirers.
Kadimastem willl now have better access to U.S. capital markets and liquidity. This is something that cannot be understated. The U.S. is the biggest market in the world by a long shot. They will undoubtably have a stronger cash runway post-merger.
NLS plans to divest certain legacy assets and distribute proceeds (via CVR) to its existing shareholders. NLS shareholders can potentially receive value from those legacy assets aside from the equity in the new entity.
Kadimastem’s lead assets like AstroRx for ALS, IsletRx for insulin-dependent diabetes have high upside potential if successful in trials.
NLSP also completed some major financing events that will propel them forward through the merger. During the first quarter of 2025, they initially closed two equity financing transactions, resulting in aggregate gross proceeds of $2.5 million, priced at $3.10 and $1.65 per share at a premium to the market share price as of the date of the transactions, representing a premium of 48% and 10%, respectively. They also signed a $25 million equity facility commitment. The potential proceeds from this facility are earmarked to support the merger with Kadimastem Ltd. and the Company’s advance clinical programs.
Since announcing its proposed merger with Kadimastem, NLS Pharmaceutics has raised over $6 million to support both the transaction itself and the combined company’s clinical trials once the merger is complete. The company has also converted all outstanding liabilities into equity, leaving it completely debt-free. These financing moves have not only strengthened the balance sheet but also extended the cash runway, giving NLS the resources to continue advancing its R&D programs while moving forward with the merger execution.
In early 2025, NLS Pharmaceutics reported encouraging preclinical data for AEX-2, further supporting the potential of its dual orexin receptor agonist (DOXA) platform in treating central nervous system disorders. Around the same time Kadimastem, together with iTolerance, successfully completed a Pre-IND meeting with the FDA for its Type 1 Diabetes therapy—an important milestone that adds a significant asset to the future combined entity, NewcelX. By April 2025, additional progress came through Study KO-943, where Mazindol ER demonstrated the ability to significantly reduce fentanyl-induced reward behaviors in animal models. These results serve as meaningful proof-points for both safety and therapeutic potential as the program advances toward next-stage trials.
Looking ahead to the anticipated closing of the merger, the combined company is preparing to advance multiple clinical milestones. These include the launch of a Phase IIa multi-site clinical trial of AstroRx® in ALS, the initiation of a Phase I study of IsletRx for Type 1 diabetes, and the continued preclinical development of AEX-2 and AEX-41 as they progress toward late-stage studies in conditions such as narcolepsy and neurodegeneration.
One of the fastest-growing sectors in global healthcare is ADHD, a market projected to reach $70+ billion by 2032. NLSP’s lead candidate, Mazindol ER, directly addresses this space — and more.
Mazindol ER: A Phase 3-Ready Asset with Broad Potential
Mazindol ER is an extended-release formulation of a well-studied compound with a proven safety profile. It’s Phase 3-ready for both:
• Attention Deficit Hyperactivity Disorder (ADHD)
• Narcolepsy and Excessive Daytime Sleepiness (EDS)
With Orphan Drug Designation in both the U.S. and Europe, and a strong Phase 2 data package in hand, Mazindol ER is positioned to move swiftly through the regulatory pipeline. The upcoming AMAZE Phase 3 trial will focus on adult patients with narcolepsy, a $4.5 billion market projected by 2027.
⸻
Strategic Expansion: Tackling the Opioid Crisis
In a significant step beyond CNS and sleep disorders, NLS also holds a U.S. patent for the use of Mazindol ER in treating opioid use disorder, including heroin and fentanyl addiction. A preclinical study targeting fentanyl addiction is underway and expected to yield promising results.
This positions NLSP within the $5 billion opioid addiction treatment market, a high-priority area for the U.S. government and public health institutions. With fentanyl-related overdoses now the leading cause of drug-related deaths in the U.S., the need for non-opioid alternatives has never been greater. Mazindol ER’s unique pharmacological profile — modulating dopaminergic and noradrenergic pathways without being addictive — makes it a compelling candidate in this space.
NLS isn’t just riding the momentum of emerging markets in ADHD and sleep disorders — they’re building long-term value through intelligent pipeline development, strategic IP, and a focus on real-world, unmet clinical needs. And now, with its entrance into the fight against opioid addiction, NLSP adds a compelling new dimension to its mission.
In a market that punishes hype and rewards substance, NLSP is on the path to quietly execute.

PIPELINE
NLS Pharmaceutics CEO Issues Letter to Shareholders
Dear Shareholders,
We are delighted to share a summary of NLS Pharmaceutics’ achieved milestones since the fourth quarter of 2024. Here’s a snapshot of our progress and what lies ahead:
1. Completion of Major Financing Events
- During the first quarter of 2025, we initially closed two equity financing transactions, resulting in aggregate gross proceeds of $2.5 million, priced at $3.10 and $1.65 per share at a premium to the market share price as of the date of the transactions, representing a premium of 48% and 10%, respectively.
- In addition, we signed a $25 million equity facility commitment. The potential proceeds from this facility are earmarked to support the merger with Kadimastem Ltd. (TASE: KDST). (“Kadimastem”) and the Company’s advance clinical programs.
Since we first announced the proposed merger with Kadimastem, we have raised more than $6 million in the aggregate to support the pending transaction and the combined company’s clinical trials post-merger. Moreover, we have converted all of the Company’s outstanding liabilities into equity, reflecting a strong balance sheet of a company free of debt. These financing events have not only strengthened our balance sheet, but have extended our cash runway, enabling us to pursue continued research & development efforts and proceed with the execution of the merger.
2. Positive Clinical and Preclinical Developments
- In February 2025, we announced encouraging preclinical data for AEX–2, further validating our dual orexin receptor agonist platform (“DOXA”) for central nervous systems (“CNS”) disorders.
- In February 2025, we announced that Kadimastem and iTolerance successfully completed a Pre-IND Meeting with the U.S. Food and Drug Administration (FDA) for its Type 1 Diabetes Treatment, representing a significant asset for the merged company, NewcelX.
- In April 2025, positive results from Study KO–943 revealed that Mazindol ER significantly reduced fentanyl–induced reward behaviors in animal models. These results represent important proof–points for both safety and therapeutic potential as we prepare for next–stage trials.
3. Merger with Kadimastem — Strategic Synergy in Action
- In January 2025, following the approval by the board of directors of NLS and Kadimastem, Kadimastem’s shareholders overwhelmingly approved the merger, a critical step in closing the merger and in bringing together the complementary strengths of Kadimastem and NLS.
- Amendments to the Registration Statements on Form F–4 were filed, paving the way for the final NLS shareholder vote.
Today, the merger process is progressing. We target completing the merger in early Q3 2025, subject to requirements set forth by all regulatory agents.
Ronen Twito, Executive Chairman and Chief Executive Officer of Kadimastem, said, “We are excited about the upcoming merger, which aligns with our strategic goals and will enable us to provide comprehensive therapeutic options for patients. By joining forces, we can better serve those with diabetes and combat its complexity. This merger demonstrates a commitment to innovation, patient care, and long-term value. Together, we aim to improve the lives of diabetics and patients with related conditions. We are committed to ensuring a smooth integration process as we move forward, following the completion of the merger. In addition to the equity investment, we believe the committed equity facility agreement of $25 million will enhance the combined company’s balance sheet and could provide flexibility to support future pipeline development.”
4. What the Combined Company Will Look Like
- The combined entity will be named NewcelX and intends to be a Nasdaq–listed clinical–stage biotech with a robust pipeline spanning neurodegenerative disease (AstroRx® for amyotrophic lateral sclerosis (“ALS”) in a Phase IIa stage), diabetes (IsletRx), and CNS disorders via the DOXA platform.
- Shareholders are expected to continue to benefit from our legacy assets—notably Mazindol ER—through contingent value rights subject to potential future sales.
5. Looking AheadAs we approach the anticipated closing of the merger, we are preparing to rapidly initiate:
- Phase IIa multi–site clinical trial for AstroRx®, targeting ALS.
- Phase I study for IsletRx in Type I diabetes.
- Continued preclinical progression of AEX–2/AEX–41 toward late–stage development (e.g., narcolepsy, neurodegeneration).
Our anticipated merger with Kadimastem is expected to mark a transformational pivot—uniting our proprietary DOXA platform and the cell–therapy assets under one roof. With a fortified capital structure, and a unified board guiding our research and development direction, we are strategically positioned for meaningful value creation.
In ConclusionThanks to your trust and resilience, NLS stands on the edge of a powerful new chapter. We anticipate the closing of the merger to occur in the near future, followed by an energized integration phase and accelerated clinical momentum.
Thank you for your enduring support. Together, we will accelerate development, diversify our pipeline, and enhance long-term shareholder value.
With optimism,Alexander C. ZwyerChief Executive OfficerNLS Pharmaceutics (NASDAQ: NLSP)
About NLS Pharmaceutics Ltd.
NLS is a global development-stage biopharmaceutical company, working with a network of world-class partners and internationally recognized scientists, focused on the discovery and development of innovative therapies for patients with rare and complex central nervous system disorders who have unmet medical needs. Headquartered in Switzerland and founded in 2015, NLS is led by an experienced management team with a track record of developing and commercializing product candidates. For more information, please visit www.nlspharma.com.
About Kadimastem
Kadimastem is a clinical stage cell therapy company, developing “off-the-shelf”, allogeneic, proprietary cell products based on its technology platform for the expansion and differentiation of Human Embryonic Stem Cells (hESCs) into functional cells. AstroRx®, the company’s lead product, is an astrocyte cell therapy in clinical development for the treatment for ALS and in pre-clinical studies for other neurodegenerative indications.
IsletRx is the company’s treatment for diabetes. IsletRx is comprised of functional pancreatic islet cells producing and releasing insulin and glucagon, intended to treat and potentially cure patients with insulin-dependent diabetes. Kadimastem was founded by Professor Michel Revel, Chief Scientific Officer of Kadimastem and Professor Emeritus of Molecular Genetics at the Weizmann Institute of Science. Professor Revel received the Israel Prize for the invention and development of Rebif®, a multiple sclerosis blockbuster drug sold worldwide. Kadimastem is traded on the Tel Aviv Stock Exchange (TASE: KDST).
MANAGEMENT TEAM

Ronen Twito CPA | Executive Chairman Nominee and CEO
Mr. Ronen Twito serves as Executive Chairman and CEO of Kadimastem, having joined the company in December 2020. Under his leadership, he successfully repositioned the company’s strategic direction, establishing Kadimastem as an emerging global leader in the cell therapy industry. Mr. Twito brings over two decades of executive leadership experience in the biotech and high-tech sectors, with a distinguished track record of driving corporate growth and maximizing shareholder value across both Nasdaq and Tel Aviv Stock Exchange listed companies.
Throughout his career, Mr. Twito has demonstrated exceptional expertise in corporate finance and strategic leadership, successfully orchestrating multiple IPOs, follow-on offerings, and high-value strategic licensing and collaboration agreements within the clinical biotech sector. His executive tenure includes pivotal roles as Deputy CEO and CFO at XTL Biopharmaceuticals (XTLB), Chief Executive Officer at Intercure Ltd, Executive Chairman at Bubbles Intergroup Ltd (BBLS), Deputy CEO and CFO at Cellect Biotechnology (APOP), External Director at MTS (MTSL), and VP Finance and Head of Israel Operations at BioBlast Pharma (ORPN). Earlier in his career, he served as Corporate Finance Director at LeadCom Integrated Solutions, a global company traded on London’s AIM market, and as Manager at Ernst & Young.
Mr. Twito is a Certified Public Accountant (CPA) in Israel and holds a bachelor’s degree in Business Administration and Accounting. He maintains active membership in the Institute of Certified Public Accountants in Israel.

Prof. Michel Revel, MD, PhD | Chief Scientist Officer and Director Nominee
Prof. Revel is Professor Emeritus of Molecular Genetics at the Weizmann Institute of Science. His research on Interferon, its mechanisms of action and the isolation of the human Interferon-beta gene, have led to the development of Interferon-beta therapy for the treatment of multiple sclerosis, Rebif®, Blockbuster drug marketed worldwide.
In recent years, Prof. Revel’s laboratory focused on hESC and succeeded to produce nerve myelinating cells that, when transplanted in myelin-deficient animals, have regenerated the myelin coating. These studies contributed to the development of a suspension culture technology for hESC which can then be used to produce differentiated human cells such as insulin-producing pancreatic beta cells and nerve myelinating cells.
Alongside his research and development activity, Prof. Revel is deeply involved in the ethics of science and biotechnology, and served as chairman of the Israel National Bioethics Council, and was a member of the International Bioethics Committee of UNESCO.
Prof. Revel was the recipient of the Israel Prize for medical research, the EMET Prize for biotechnology, and is a member of the Israel Academy of Science and Humanities. He has been a member of Israel’s National Committee for Biotechnology, serving for three years as its chairman.
CLICK HERE FOR Prof. Michel Revel full CURRICULUM VITAE

Kfir Molakandov PhD | VP Research and Development
Dr. Kfir Molakandov started his academic career in developmental biology with Prof. Sarah Ferber’s research on transdifferentiation of liver to the pancreas.
During his PhD, he trained at the Gene Therapy Center, Alabama, USA, and specialized in adenoviral vectors construction, modifications and purification. Following the completion of his thesis, Dr. Molakandov joined Kadimastem.
He leads the team that is developing cell-therapy for diabetes using Pluripotent Stem Cells as the cell source. His team has dedicated the last few years in developing a large scale robust protocol for generating islet cells in high quantities and high quality.
Currently, we have finalized the production process that includes a full characterization of the different stages of differentiation using molecular tools and physiological parameters.
Accordingly, we are now moving towards the clinical application of this technology, aiming to bring these therapeutic cells to clinical trials.

Ariel Revel, MD, | Director of Medical Affairs
Prof. Ariel Revel, MD, is a professor of medicine at Tel Aviv University and a Visiting Professor at Oxford University in the UK and Stanford University in California. He is among Israel’s leading specialists in gynecology and obstetrics and has for many years been involved in IVF and minimally invasive surgery.
Ariel Revel served as a battalion and brigade physician, and raised his family in Nataf. He volunteers with Etgarim, where he kayaks and rides tandem bikes with children and teens with disabilities, and with Physicians for Human Rights, as a physician at the clinic for status-less individuals in Jaffa.
He is a an important figure on the medical landscape, and this together with his impressive knowledge and many years of experience has produced one of the most fascinating books published in recent years in Israel.
Ariel at Kadimastem is responsible for the following areas
1) To represent the surgical and medical aspects of islet transplantation for patients with diabetes.
2) To promote to medical doctors that cell therapy is the future of medicine. To improve and perhaps cure neurodegenerative diseases such as ALS.
3) Present to general public and potential inventors the potential of human embryonic stem cells to produce clinical level adult cells to cure Diabetes, Glaucoma and Traumatic Spinal Cord Injuries
NEWS
Sep 10, 2025
Sep 4, 2025
Jul 17, 2025
NLS Pharmaceutics and Kadimastem Announce Pricing and Closing of $1 Million Equity Financing
Jun 30, 2025
NLS Pharmaceutics CEO Issues Letter to Shareholders
Jun 16, 2025
May 21, 2025
Apr 15, 2025
Mar 31, 2025
NLS Pharmaceutics CEO Issues Letter to Shareholders
Mar 10, 2025
Feb 27, 2025
Feb 25, 2025
Feb 25, 2025
Feb 10, 2025
Kadimastem Shareholders Approved the Merger with NLS Pharmaceutics
Jan 31, 2025
Jan 30, 2025
Jan 28, 2025
Kadimastem Calls for a Special General Meeting of Shareholders to Approve the Merger with NLS
Jan 16, 2025
Jan 8, 2025
Dec 30, 2024
Dec 19, 2024
SINCERELY,

DISCLAIMER
THIS WEBSITE/NEWSLETTER IS OWNED SUBSIDIARY BY DEDICATED INVESTORS, LLC.
OUR REPORTS/RELEASES ARE A COMMERCIAL ADVERTISEMENT AND ARE FOR GENERAL INFORMATION PURPOSES ONLY. WE ARE ENGAGED IN THE BUSINESS OF MARKETING AND ADVERTISING COMPANIES FOR MONETARY COMPENSATION. WE HAVE BEEN COMPENSATED A FEE OF SEVENTEEN THOUSAND FIVE HUNDRED USD BY LFG EQUITIES CORP FOR A ONE DAY NLSP AWARENESS CAMPAIGN. NEVER INVEST IN ANY STOCK FEATURED ON OUR SITE OR EMAILS UNLESS YOU CAN AFFORD TO LOSE YOUR ENTIRE INVESTMENT. THE DISCLAIMER IS TO BE READ AND FULLY UNDERSTOOD BEFORE USING OUR SERVICES, JOINING OUR SITE OR OUR EMAIL/BLOG LIST AS WELL AS ANY SOCIAL NETWORKING PLATFORMS WE MAY USE.PLEASE NOTE WELL: DEDICATED INVESTORS LLC AND ITS EMPLOYEES ARE NOT A REGISTERED INVESTMENT ADVISOR, BROKER DEALER OR A MEMBER OF ANY ASSOCIATION FOR OTHER RESEARCH PROVIDERS IN ANY JURISDICTION WHATSOEVER.RELEASE OF LIABILITY: THROUGH USE OF THIS WEBSITE VIEWING OR USING YOU AGREE TO HOLD DEDICATED INVESTORS LLC, ITS OPERATORS OWNERS AND EMPLOYEES HARMLESS AND TO COMPLETELY RELEASE THEM FROM ANY AND ALL LIABILITY DUE TO ANY AND ALL LOSS (MONETARY OR OTHERWISE), DAMAGE (MONETARY OR OTHERWISE), OR INJURY (MONETARY OR OTHERWISE) THAT YOU MAY INCUR. THE INFORMATION CONTAINED HEREIN IS BASED ON SOURCES WHICH WE BELIEVE TO BE RELIABLE BUT IS NOT GUARANTEED BY US AS BEING ACCURATE AND DOES NOT PURPORT TO BE A COMPLETE STATEMENT OR SUMMARY OF THE AVAILABLE DATA. DEDICATED INVESTORS LLC ENCOURAGES READERS AND INVESTORS TO SUPPLEMENT THE INFORMATION IN THESE REPORTS WITH INDEPENDENT RESEARCH AND OTHER PROFESSIONAL ADVICE. ALL INFORMATION ON FEATURED COMPANIES IS PROVIDED BY THE COMPANIES PROFILED, OR IS AVAILABLE FROM PUBLIC SOURCES AND DEDICATED INVESTORS LLC MAKES NO REPRESENTATIONS, WARRANTIES OR GUARANTEES AS TO THE ACCURACY OR COMPLETENESS OF THE DISCLOSURE BY THE PROFILED COMPANIES. NONE OF THE MATERIALS OR ADVERTISEMENTS HEREIN CONSTITUTE OFFERS OR SOLICITATIONS TO PURCHASE OR SELL SECURITIES OF THE COMPANIES PROFILED HEREIN AND ANY DECISION TO INVEST IN ANY SUCH COMPANY OR OTHER FINANCIAL DECISIONS SHOULD NOT BE MADE BASED UPON THE INFORMATION PROVIDED HEREIN. INSTEAD DEDICATED INVESTORS LLC STRONGLY URGES YOU CONDUCT A COMPLETE AND INDEPENDENT INVESTIGATION OF THE RESPECTIVE COMPANIES AND CONSIDERATION OF ALL PERTINENT RISKS. READERS ARE ADVISED TO REVIEW SEC PERIODIC REPORTS: FORMS 10-Q, 10K, FORM 8-K, INSIDER REPORTS, FORMS 3, 4, 5 SCHEDULE 13D.DEDICATED INVESTORS LLC IS COMPLIANT WITH THE CAN SPAM ACT OF 2003. DEDICATED INVESTORS LLC DOES NOT OFFER SUCH ADVICE OR ANALYSIS, AND DEDICATED INVESTORS LLC FURTHER URGES YOU TO CONSULT YOUR OWN INDEPENDENT TAX, BUSINESS, FINANCIAL AND INVESTMENT ADVISORS. INVESTING IN MICRO-CAP AND GROWTH SECURITIES IS HIGHLY SPECULATIVE AND CARRIES AND EXTREMELY HIGH DEGREE OF RISK. IT IS POSSIBLE THAT AN INVESTORS INVESTMENT MAY BE LOST OR IMPAIRED DUE TO THE SPECULATIVE NATURE OF THE COMPANIES PROFILED.THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 PROVIDES INVESTORS A SAFE HARBOR IN REGARD TO FORWARD-LOOKING STATEMENTS. ANY STATEMENTS THAT EXPRESS OR INVOLVE DISCUSSIONS WITH RESPECT TO PREDICTIONS, EXPECTATIONS, BELIEFS, PLANS, PROJECTIONS, OBJECTIVES, GOALS, ASSUMPTIONS OR FUTURE EVENTS OR PERFORMANCE ARE NOT STATEMENTS OF HISTORICAL FACT MAY BE FORWARD LOOKING STATEMENTS. FORWARD LOOKING STATEMENTS ARE BASED ON EXPECTATIONS, ESTIMATES, AND PROJECTIONS AT THE TIME THE STATEMENTS ARE MADE THAT INVOLVE A NUMBER OF RISKS AND UNCERTAINTIES WHICH COULD CAUSE ACTUAL RESULTS OR EVENTS TO DIFFER MATERIALLY FROM THOSE PRESENTLY ANTICIPATED. FORWARD LOOKING STATEMENTS IN THIS ACTION MAY BE IDENTIFIED THROUGH USE OF WORDS SUCH AS PROJECTS, FORESEE, EXPECTS, WILL, ANTICIPATES, ESTIMATES, BELIEVES, UNDERSTANDS, OR THAT BY STATEMENTS INDICATING CERTAIN ACTIONS & QUOTE; MAY, COULD, OR MIGHT OCCUR. UNDERSTAND THERE IS NO GUARANTEE PAST PERFORMANCE WILL BE INDICATIVE OF FUTURE RESULTS. IN PREPARING THIS PUBLICATION, DEDICATED INVESTORS LLC HAS RELIED UPON INFORMATION SUPPLIED BY ITS CUSTOMERS, PUBLICLY AVAILABLE INFORMATION AND PRESS RELEASES WHICH IT BELIEVES TO BE RELIABLE; HOWEVER, SUCH RELIABILITY CANNOT BE GUARANTEED. INVESTORS SHOULD NOT RELY ON THE INFORMATION CONTAINED IN THIS WEBSITE. RATHER, INVESTORS SHOULD USE THE INFORMATION CONTAINED IN THIS WEBSITE AS A STARTING POINT FOR DOING ADDITIONAL INDEPENDENT RESEARCH ON THE FEATURED COMPANIES. THE ADVERTISEMENTS IN THIS WEBSITE ARE BELIEVED TO BE RELIABLE, HOWEVER, DEDICATED INVESTORS LLC AND ITS OWNERS, AFFILIATES, SUBSIDIARIES, OFFICERS, DIRECTORS, REPRESENTATIVES AND AGENTS DISCLAIM ANY LIABILITY AS TO THE COMPLETENESS OR ACCURACY OF THE INFORMATION CONTAINED IN ANY ADVERTISEMENT AND FOR ANY OMISSIONS OF MATERIALS FACTS FROM SUCH ADVERTISEMENT. DEDICATED INVESTORS LLC IS NOT RESPONSIBLE FOR ANY CLAIMS MADE BY THE COMPANIES ADVERTISED HEREIN, NOR IS DEDICATED INVESTORS LLC RESPONSIBLE FOR ANY OTHER PROMOTIONAL FIRM, ITS PROGRAM OR ITS STRUCTURE. DEDICATED INVESTORS LLC IS NOT AFFILIATED WITH ANY EXCHANGE, ELECTRONIC QUOTATION SYSTEM, THE SECURITIES EXCHANGE COMMISSION OR FINRA.
