**Sponsored by LFG Equities Corp.

GeoVax is stepping into 2026 with something it hasn’t had before: a clear, executable path to real commercial relevance.
READ THE INVESTOR PRESENTATION HERE
_________________________
Hello Everyone,
We have a new company that just experienced several events that make it extremely important that we put this one on your radar immediately.
Pull up GeoVax Labs (NASDAQ: GOVX) right away.
GeoVax is stepping into 2026 with something it hasn’t had before: a clear, executable path to real commercial relevance. After years of platform building, the story is no longer theoretical — the company now has a defined late-stage program, manufacturing ready to scale, and multiple clinical readouts that could reshape how the market values the pipeline. The centerpiece is its Mpox/smallpox vaccine, which quietly received regulatory alignment in Europe for a single pivotal Phase 3 immunobridging study — a faster, cheaper, and far less risky route to approval than a traditional efficacy trial. In a world where governments are actively trying to secure domestic vaccine supply and reduce reliance on a single overseas producer, that positioning matters. Layer on top the potential for U.S.-based continuous manufacturing, fresh Phase 2 data in its next-generation COVID program targeting immunocompromised patients, and the move to push Gedeptin into a checkpoint-inhibitor combination study in oncology, and you get a company transitioning from “science project” to “catalyst cycle.”
The next twelve months aren’t about promises — they’re about execution: trial initiations, data, partnerships, and funding decisions that will determine whether this becomes a procurement-driven revenue story or just another development-stage biotech. For the first time, the pieces are lining up at the same time, and that convergence is what makes this GOVX genuinely worth watching as we kick off 2026.
There are several recent events that make GOVX enticing to research right now. We know that timing is everything in the markets whether its Gold, BTC, Commodities or equities. We are looking this one as it is just coming off of a restructuring which dramatically reduced the float on GOVX. Finviz has it under 1.75 Million right now.
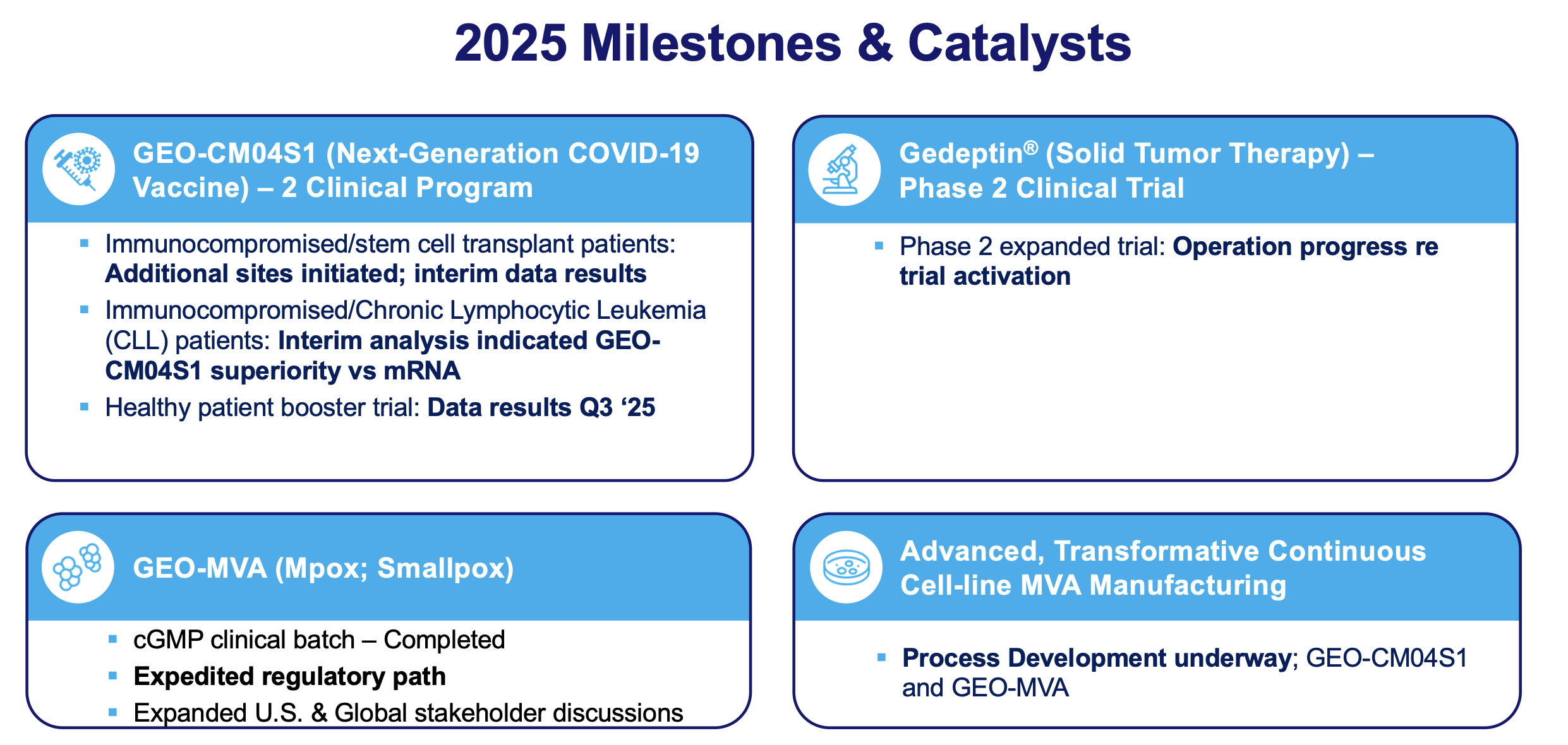
They also just announced the successful completion of fill-finish for the initial clinical batch of GEO-MVA, its next-generation Mpox/smallpox vaccine. The product has now entered final release evaluation, the concluding quality-control and compliance process required before shipment for clinical use, positioning the Company for Phase 3 immunobridging trial start-up activities in Q1 2026.
Clinical and Regulatory Milestone
Fill-finish – the sterile, cGMP-regulated process of filling, sealing, and packaging vaccine vials – marks the last manufacturing step before a vaccine may enter clinical study supply channels. With fill-finish complete and GEO-MVA now undergoing final release evaluation, GeoVax has moved into the final pre-clinical-deployment phase of its EMA-aligned clinical program.
In June 2025, the European Medicines Agency (EMA) Scientific Advice confirmed that a single Phase 3 immunobridging study demonstrating immune comparability to the approved MVA vaccine, Imvanex®, would be sufficient to evaluate GEO-MVA’s efficacy. This provides a clear, accelerated regulatory path to licensure.
This milestone coincides with increasing Mpox activity globally – including expanding Clade I outbreaks in Africa and emerging cases in the United States – exposing vulnerabilities associated with global dependence on a sole foreign MVA vaccine supplier. GEO-MVA is designed to expand supply, diversify sources, and strengthen biodefense infrastructure.
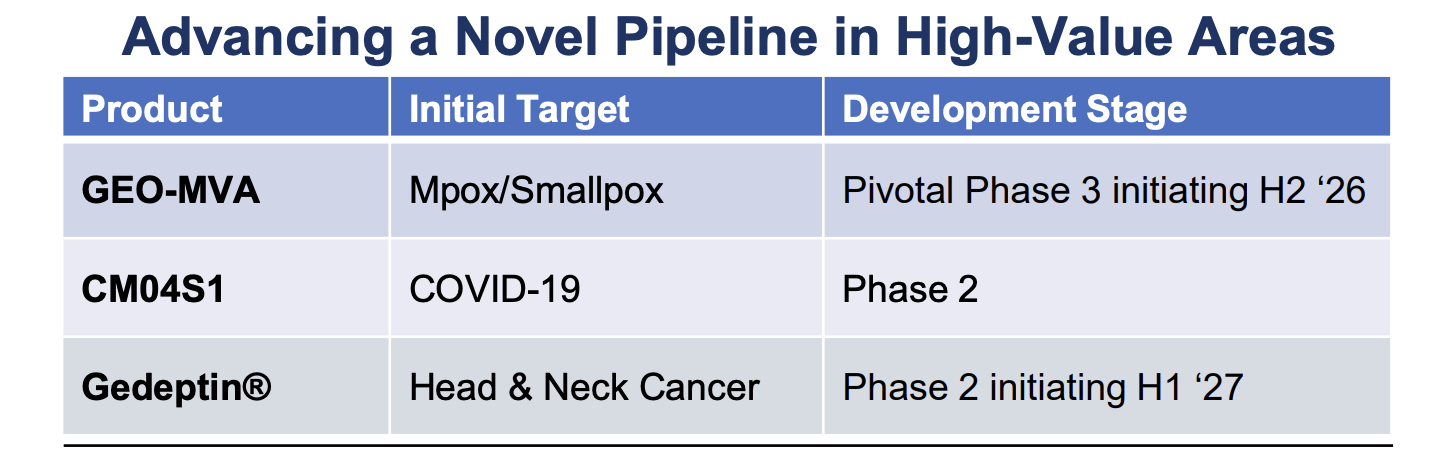
The clinical-stage biotechnology company developing novel vaccines for many of the world’s most threatening infectious diseases and therapies for solid tumor cancers. The company’s lead clinical program is GEO-CM04S1, a next-generation COVID-19 vaccine for which GeoVax was recently awarded a BARDA-funded contract to sponsor a 10,000-participant Phase 2b clinical trial to evaluate the efficacy of GEO-CM04S1 versus an approved COVID-19 vaccine. In addition, GEO-CM04S1 is currently in three Phase 2 clinical trials, being evaluated as (1) a primary vaccine for immunocompromised patients such as those suffering from hematologic cancers and other patient populations for whom the current authorized COVID-19 vaccines are insufficient, (2) a booster vaccine in patients with chronic lymphocytic leukemia (CLL) and (3) a more robust, durable COVID-19 booster among healthy patients who previously received the mRNA vaccines. In oncology the lead clinical program is evaluating a novel oncolytic solid tumor gene-directed therapy, Gedeptin®, having recently completed a multicenter Phase 1/2 clinical trial for advanced head and neck cancers. A Phase 2 clinical trial in first recurrent head and neck cancer, evaluating Gedeptin® combined with an immune checkpoint inhibitor is planned to initiate during the first half of 2025. GeoVax has a strong IP portfolio in support of its technologies and product candidates, holding worldwide rights for its technologies and products. The Company has a leadership team who have driven significant value creation across multiple life science companies over the past several decades.

The market is massive and the prospects are enormous.
Recently, Bavarian Nordic, a European vaccine maker of Mpox and Small Pox vaccines was offered $3B in a private equity buyout offer, which looks to be moving forward. In 2024, their MVA vaccine program generated $462M in revenue. This is not suggesting GeoVax will get a $3B buyout also, but is definitely indicative of the large potential of the market GeoVax is addressing and the strong interest from large industry players/investors.
GeoVax’s GEO-MVA candidate leverages a proprietary MVA vector to address the Mpox crisis and the ongoing threat of smallpox, offering broad protection, scalability, and resilience in vaccine supply. Unlike Bavarian Nordic, GeoVax is based in the U.S. and has invested heavily in a continuous cell line manufacturing process that supports large-scale, rapid, and cost-efficient production—an upgrade over traditional egg-based methods. This positions GeoVax to supply not only domestic needs but also bolster global pandemic preparedness and biosecurity, as recognized by recent favorable regulatory guidance from the European Medicines Agency (EMA) permitting a fast-tracked pathway to approval.
GeoVax Announces Issuance of U.S. Patent Covering Enhanced Therapeutic Use of Gedeptin(R) Gene Therapy
Patent Protects Novel Application of Gedeptin Therapy Across Multiple Solid Tumor Types; Supports Expansion of Gedeptin Product Platform
ATLANTA, GA – December 9, 2025 (NEWMEDIAWIRE) – GeoVax Labs, Inc. (Nasdaq: GOVX), a clinical-stage biotechnology company developing multi-antigen vaccines and immunotherapies against cancers and infectious diseases, today announced the issuance of U.S. Patent No. 12,453,760, titled “Enhanced Therapeutic Usage of a Purine Nucleoside Phosphorylase or Nucleoside Hydrolase Prodrug”, by the United States Patent and Trademark Office (USPTO). The patent provides composition-of-matter and method-of-use protection for GeoVax’s Gedeptin® platform in combination with targeted delivery approaches for solid tumors, including head and neck cancer.
The newly issued patent, which extends through 2045, enhances the Company’s intellectual property estate for Gedeptin and its use across a range of solid tumor cancers – consolidating GeoVax’s leadership in the field of targeted gene therapies and supporting ongoing clinical development plans.
“The issuance of this patent marks an important milestone in the advancement and protection of GeoVax’s oncology pipeline,” said David A. Dodd, Chairman and Chief Executive Officer of GeoVax. “It underscores our commitment to progressing Gedeptin, both as a monotherapy and in synergistic combination with other oncology treatment approaches as we work to deliver meaningful treatment options for patients with difficult-to-treat solid tumors.”
The Company is actively preparing for a Phase 2 clinical trial evaluating Gedeptin as a first-line therapy in combination with pembrolizumab (Keytruda®) in resectable head and neck cancer, in line with the recent shift toward neoadjuvant checkpoint strategies. Additional preclinical programs are assessing Gedeptin across other tumor types, including breast and cutaneous cancers.
About Gedeptin®
Gedeptin is a gene-directed enzyme prodrug therapy (GDEPT) designed for targeted use in solid tumors. Delivered via a non-replicating adenoviral vector encoding purine nucleoside phosphorylase (PNP) and followed by systemic fludarabine, Gedeptin generates localized cytotoxic activity within tumors while minimizing systemic toxicity. The therapy has demonstrated safety and disease control in a multi-center Phase 1/2 trial in patients with advanced head and neck cancer and has received FDA Orphan Drug Designation for oral and pharyngeal cancers.
GeoVax plans to advance Gedeptin into a Phase 2 trial in combination with pembrolizumab (Keytruda®) as a neoadjuvant regimen for resectable head and neck squamous cell carcinoma, supported by recent clinical data validating the role of immune checkpoint inhibitors in perioperative settings. Additional preclinical work is underway to assess Gedeptin combinations across other solid tumors.
Key Advantages of Gedeptin
- Localized, tumor-selective cytotoxicity
- Tumor agnostic – expansion potential across multiple solid tumors
- Synergistic potential with checkpoint inhibitors
- Favorable safety profile and orphan drug designation
- Strong patent protection through 2045
GeoVax Announces British Journal of Haematology Publication Highlighting Superior T-Cell Responses of GEO-CM04S1 in CLL Patients
DSMB Ends Comparator Arm; Trial Proceeds Exclusively With GEO-CM04S1 Following mRNA Vaccine’s Failure to Meet Primary Endpoint
Phase 2 Data Reinforce GEO-CM04S1 as an Important Next-generation Vaccine Candidate for the 40 Million U.S. and 400 Million Global Immunocompromised Patients Underserved by First-generation COVID-19 Vaccines
ATLANTA, GA – December 15, 2025 (NEWMEDIAWIRE) – GeoVax Labs, Inc. (Nasdaq: GOVX), a clinical-stage biotechnology company developing multi-antigen vaccines and immunotherapies, today announced the publication of interim Phase 2 clinical data on its next-generation COVID-19 vaccine in patients with chronic lymphocytic leukemia (CLL).
The Research Letter in the British Journal of Haematology reports that GEO-CM04S1 met the study’s primary immunologic endpoint, generating significantly stronger and more durable SARS-CoV-2-specific T-cell responses than BNT162b2 (Pfizer-BioNTech) in patients with chronic lymphocytic leukemia (CLL) – a population known for poor vaccine responsiveness.
Importantly, following interim analysis, the trial’s Data and Safety Monitoring Board (DSMB) ruled to discontinue the randomized, double-blind comparator arm after the mRNA vaccine failed to meet the predefined primary immunogenicity endpoint. Enrollment is now proceeding exclusively in a single-arm cohort receiving GEO-CM04S1, as previously described in GeoVax’s clinical update at the European Hematology Association (EHA) 2025 Conference.
GEO-CM04S1’s superior performance in enhancing cellular immune response against SARS-CoV-2 in individuals with CLL, a patient population that generally responds sub optimally to vaccines designed to induce humoral (antibody) responses, underscores its potential to fill a protection gap for profoundly immunocompromised patients. More than 40 million adults in the U.S. and 400 million globally have some degree of compromised immunity, many of whom fail to mount meaningful responses to currently authorized COVID-19 vaccines. GEO-CM04S1 is specifically designed to address this gap through its dual-antigen (Spike + Nucleocapsid), MVA-based platform, which promotes robust, durable T-cell responses that are less impacted by immune dysfunction and viral variation.
Phase 2 Study Overview (NCT05672355)
- CLL patients previously vaccinated with mRNA vaccines
- 31 enrolled; 27 evaluable for primary analysis
- Primary endpoint: greater than or equal to 3-fold rise in antigen-specific IFN-y-secreting T cells at Day 56
- Assessments: T-cell responses, binding/neutralizing antibodies, safety
- No Grade greater than or equal to 3 adverse events reported
Key Findings Published in BJH
1. GEO-CM04S1 achieved superior T-cell responses
- 40% of GEO-CM04S1 recipients met the primary endpoint vs. 14.3% for BNT162b2
- Higher Spike-specific IFN-y responses at Days 28, 56, and 84
2. Durable activation of Nucleocapsid-specific T cells
- Approximately 10-fold higher N-specific CD4 T-cell activation vs. BNT162b2
- Responses maintained through Day 180
3. Broader immune engagement in spite of CLL-associated humoral defects
- GEO-CM04S1 generated sustained N-IgG and a correlation between N-specific antibodies and T-cell activation
- mRNA vaccination produced higher early RBD-IgG titers but limited cellular immunity
Kelly T. McKee, MD, MPH, Chief Medical Officer, stated: “These results demonstrate GEO-CM04S1’s ability to address the immune limitations of CLL patients by inducing strong, durable T-cell responses to both spike and nucleocapsid proteins of SARS-CoV-2. The DSMB’s decision to discontinue the comparator arm further validates the vaccine’s clinical relevance for immunocompromised individuals.”
David Dodd, Chairman & CEO, added: “With more than 40 million immunocompromised Americans, many of whom lack durable protection from first-generation vaccines, GEO-CM04S1 represents a purpose-built solution for high-risk patients. This peer-reviewed publication strengthens our regulatory and partnering strategy as we advance toward potential commercialization.”
Medical and Commercial Significance
The findings published in BJH, combined with the DSMB’s action, reinforce the value of the differentiated profile of GEO-CM04S1 across multiple dimensions:
- Critical unmet need: Immunocompromised individuals remain vulnerable and, in many cases, sub optimally protected from the threat of SARS-CoV-2.
- Multi-antigen design: GEO-CM04S1’s dual-antigen design stimulates immune responses that appear to be more durable and variant-resilient than single-antigen mRNA approaches.
- These segments represent a $30B+ annual potential commercial market.
About GEO-CM04S1
GEO-CM04S1 is a dual-antigen MVA-vectored COVID-19 vaccine being evaluated in multiple Phase 2 trials, including a primary vaccination for immunocompromised individuals, and a booster vaccination for CLL patients.
The vaccine has generated robust immune responses in difficult-to-vaccinate populations including CAR-T and stem-cell transplant recipients, who typically fail to respond well to first-generation vaccines.

MVA TECHNOLOGY OVERVIEW
GeoVax’s vaccines are constructed to induce broader immunity through inclusion of multiple antigens into a single virus/vaccine platform. This is possible through the use of the company’s MVA vaccine platform, a large virus capable of incorporating multiple antigens into a vaccine platform.
Utilizing MVA, as a vaccine vector, allows for the targeting of multiple sites on a pathogen or cancer cell. Doing this is intended to result in a more robust and durable protective immune response. In addition, using MVA as a vaccine platform allows for the construction of vaccines which are capable of generating virus-like particles (VLPs) in the person receiving the vaccine.
The production of VLPs in the person being vaccinated is intended to mimic viral production that occurs in a natural infection, stimulating both the humoral (antibody) and cellular (T-cell) arms of the immune system to recognize, prevent, and control future infections.
MVA vectored vaccines can elicit durable (long-acting) immune responses while also possessing an excellent safety profile. MVA-VLP vaccines are designed to mimic authentic viruses in form but are not infectious or capable of replicating. As a result, VLPs can cause the body’s immune system to recognize and kill targeted infectious agents to prevent an infection or can be designed to target cancerous cells resulting in inhibited growth or destruction of tumors. VLPs can also train the immune system to recognize and kill virus-infected cells to control infection and reduce the length and severity of disease.
GEDEPTIN TECHNOLOGY OVERVIEW
A Phase 1/2 trial (NCT03754933), evaluating the safety and efficacy of repeat cycles of Gedeptin therapy in patients with recurrent head and neck squamous cell carcinoma (HNSCC), with tumor(s) accessible for injection and no curable treatment options recently completed enrollment at the Stanford University Cancer Institute, the Emory University Winship Cancer Institute, and the Thomas Jefferson University Sidney Kimmel Cancer Center.
The trial design involved repeat administration using Gedeptin followed by systemic fludarabine (prodrug). Expansion towards a larger, Phase 2 patient trial is anticipated. The FDA has granted Gedeptin orphan drug status for the intra-tumoral treatment of anatomically accessible oral and pharyngeal cancers, including cancers of the lip, tongue, gum, floor of mouth, salivary gland and other oral cavities. Also, the initial Phase 1/2 clinical study was funded by the FDA pursuant to its Orphan Products Clinical Trials Grants Program.

NEWS
GeoVax Labs Announces $1 Million Registered Direct Offering Priced At-The-Market Under Nasdaq Rules
1 hour ago
GeoVax Highlights 2026 as a Pivotal Year for Progress
Jan 20, 2026
Jan 8, 2026
Jan 6, 2026
GeoVax to Raise Approximately $3.2 Million of Gross Proceeds in Public Offering
Dec 19, 2025
Dec 18, 2025
Dec 18, 2025
Dec 17, 2025
GeoVax Receives U.S. Patent Office Notice of Allowance for Broad-Spectrum COVID-19 Vaccine Design
Dec 16, 2025
Dec 15, 2025
GeoVax Addresses Identification of New Mpox Variant
Dec 11, 2025
GeoVax Announces Addition of Renowned Global Experts to Its Scientific Advisory Board
Dec 10, 2025
Dec 9, 2025
Dec 8, 2025
GeoVax to Present at the Noble Capital Markets Twenty-First Annual Emerging Growth Equity Conference
Nov 24, 2025
GeoVax Reports Third Quarter 2025 Financial Results and Provides Business Update
Nov 13, 2025
GeoVax Recognizes World Immunization Day: Advancing Innovation and Trust in Vaccination
Nov 10, 2025
GeoVax Recognizes World Immunization Day: Advancing Innovation and Trust in Vaccination
Nov 10, 2025
Nov 4, 2025
Nov 3, 2025
MANAGEMENT




SINCERELY,

DISCLAIMER
THIS WEBSITE/NEWSLETTER IS OWNED SUBSIDIARY BY DEDICATED INVESTORS, LLC.
OUR REPORTS/RELEASES ARE A COMMERCIAL ADVERTISEMENT AND ARE FOR GENERAL INFORMATION PURPOSES ONLY. WE ARE ENGAGED IN THE BUSINESS OF MARKETING AND ADVERTISING COMPANIES FOR MONETARY COMPENSATION. WE HAVE BEEN COMPENSATED A FEE OF FIFTEEN THOUSAND USD BY LFG EQUITIES CORP FOR A ONE DAY GOVX AWARENESS CAMPAIGN. NEVER INVEST IN ANY STOCK FEATURED ON OUR SITE OR EMAILS UNLESS YOU CAN AFFORD TO LOSE YOUR ENTIRE INVESTMENT. THE DISCLAIMER IS TO BE READ AND FULLY UNDERSTOOD BEFORE USING OUR SERVICES, JOINING OUR SITE OR OUR EMAIL/BLOG LIST AS WELL AS ANY SOCIAL NETWORKING PLATFORMS WE MAY USE.PLEASE NOTE WELL: DEDICATED INVESTORS LLC AND ITS EMPLOYEES ARE NOT A REGISTERED INVESTMENT ADVISOR, BROKER DEALER OR A MEMBER OF ANY ASSOCIATION FOR OTHER RESEARCH PROVIDERS IN ANY JURISDICTION WHATSOEVER.RELEASE OF LIABILITY: THROUGH USE OF THIS WEBSITE VIEWING OR USING YOU AGREE TO HOLD DEDICATED INVESTORS LLC, ITS OPERATORS OWNERS AND EMPLOYEES HARMLESS AND TO COMPLETELY RELEASE THEM FROM ANY AND ALL LIABILITY DUE TO ANY AND ALL LOSS (MONETARY OR OTHERWISE), DAMAGE (MONETARY OR OTHERWISE), OR INJURY (MONETARY OR OTHERWISE) THAT YOU MAY INCUR. THE INFORMATION CONTAINED HEREIN IS BASED ON SOURCES WHICH WE BELIEVE TO BE RELIABLE BUT IS NOT GUARANTEED BY US AS BEING ACCURATE AND DOES NOT PURPORT TO BE A COMPLETE STATEMENT OR SUMMARY OF THE AVAILABLE DATA. DEDICATED INVESTORS LLC ENCOURAGES READERS AND INVESTORS TO SUPPLEMENT THE INFORMATION IN THESE REPORTS WITH INDEPENDENT RESEARCH AND OTHER PROFESSIONAL ADVICE. ALL INFORMATION ON FEATURED COMPANIES IS PROVIDED BY THE COMPANIES PROFILED, OR IS AVAILABLE FROM PUBLIC SOURCES AND DEDICATED INVESTORS LLC MAKES NO REPRESENTATIONS, WARRANTIES OR GUARANTEES AS TO THE ACCURACY OR COMPLETENESS OF THE DISCLOSURE BY THE PROFILED COMPANIES. NONE OF THE MATERIALS OR ADVERTISEMENTS HEREIN CONSTITUTE OFFERS OR SOLICITATIONS TO PURCHASE OR SELL SECURITIES OF THE COMPANIES PROFILED HEREIN AND ANY DECISION TO INVEST IN ANY SUCH COMPANY OR OTHER FINANCIAL DECISIONS SHOULD NOT BE MADE BASED UPON THE INFORMATION PROVIDED HEREIN. INSTEAD DEDICATED INVESTORS LLC STRONGLY URGES YOU CONDUCT A COMPLETE AND INDEPENDENT INVESTIGATION OF THE RESPECTIVE COMPANIES AND CONSIDERATION OF ALL PERTINENT RISKS. READERS ARE ADVISED TO REVIEW SEC PERIODIC REPORTS: FORMS 10-Q, 10K, FORM 8-K, INSIDER REPORTS, FORMS 3, 4, 5 SCHEDULE 13D.DEDICATED INVESTORS LLC IS COMPLIANT WITH THE CAN SPAM ACT OF 2003. DEDICATED INVESTORS LLC DOES NOT OFFER SUCH ADVICE OR ANALYSIS, AND DEDICATED INVESTORS LLC FURTHER URGES YOU TO CONSULT YOUR OWN INDEPENDENT TAX, BUSINESS, FINANCIAL AND INVESTMENT ADVISORS. INVESTING IN MICRO-CAP AND GROWTH SECURITIES IS HIGHLY SPECULATIVE AND CARRIES AND EXTREMELY HIGH DEGREE OF RISK. IT IS POSSIBLE THAT AN INVESTORS INVESTMENT MAY BE LOST OR IMPAIRED DUE TO THE SPECULATIVE NATURE OF THE COMPANIES PROFILED.THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 PROVIDES INVESTORS A SAFE HARBOR IN REGARD TO FORWARD-LOOKING STATEMENTS. ANY STATEMENTS THAT EXPRESS OR INVOLVE DISCUSSIONS WITH RESPECT TO PREDICTIONS, EXPECTATIONS, BELIEFS, PLANS, PROJECTIONS, OBJECTIVES, GOALS, ASSUMPTIONS OR FUTURE EVENTS OR PERFORMANCE ARE NOT STATEMENTS OF HISTORICAL FACT MAY BE FORWARD LOOKING STATEMENTS. FORWARD LOOKING STATEMENTS ARE BASED ON EXPECTATIONS, ESTIMATES, AND PROJECTIONS AT THE TIME THE STATEMENTS ARE MADE THAT INVOLVE A NUMBER OF RISKS AND UNCERTAINTIES WHICH COULD CAUSE ACTUAL RESULTS OR EVENTS TO DIFFER MATERIALLY FROM THOSE PRESENTLY ANTICIPATED. FORWARD LOOKING STATEMENTS IN THIS ACTION MAY BE IDENTIFIED THROUGH USE OF WORDS SUCH AS PROJECTS, FORESEE, EXPECTS, WILL, ANTICIPATES, ESTIMATES, BELIEVES, UNDERSTANDS, OR THAT BY STATEMENTS INDICATING CERTAIN ACTIONS & QUOTE; MAY, COULD, OR MIGHT OCCUR. UNDERSTAND THERE IS NO GUARANTEE PAST PERFORMANCE WILL BE INDICATIVE OF FUTURE RESULTS. IN PREPARING THIS PUBLICATION, DEDICATED INVESTORS LLC HAS RELIED UPON INFORMATION SUPPLIED BY ITS CUSTOMERS, PUBLICLY AVAILABLE INFORMATION AND PRESS RELEASES WHICH IT BELIEVES TO BE RELIABLE; HOWEVER, SUCH RELIABILITY CANNOT BE GUARANTEED. INVESTORS SHOULD NOT RELY ON THE INFORMATION CONTAINED IN THIS WEBSITE. RATHER, INVESTORS SHOULD USE THE INFORMATION CONTAINED IN THIS WEBSITE AS A STARTING POINT FOR DOING ADDITIONAL INDEPENDENT RESEARCH ON THE FEATURED COMPANIES. THE ADVERTISEMENTS IN THIS WEBSITE ARE BELIEVED TO BE RELIABLE, HOWEVER, DEDICATED INVESTORS LLC AND ITS OWNERS, AFFILIATES, SUBSIDIARIES, OFFICERS, DIRECTORS, REPRESENTATIVES AND AGENTS DISCLAIM ANY LIABILITY AS TO THE COMPLETENESS OR ACCURACY OF THE INFORMATION CONTAINED IN ANY ADVERTISEMENT AND FOR ANY OMISSIONS OF MATERIALS FACTS FROM SUCH ADVERTISEMENT. DEDICATED INVESTORS LLC IS NOT RESPONSIBLE FOR ANY CLAIMS MADE BY THE COMPANIES ADVERTISED HEREIN, NOR IS DEDICATED INVESTORS LLC RESPONSIBLE FOR ANY OTHER PROMOTIONAL FIRM, ITS PROGRAM OR ITS STRUCTURE. DEDICATED INVESTORS LLC IS NOT AFFILIATED WITH ANY EXCHANGE, ELECTRONIC QUOTATION SYSTEM, THE SECURITIES EXCHANGE COMMISSION OR FINRA.
